Cellular Respiration and Fermentation
Here’s a song about cellular respiration.
Fermentation:
Lactic acid fermentation is done by some fungi, some
bacteria like theLactobacillus
acidophilus. in yogurt, and sometimes by our muscles. Normally our
muscles do cellular respiration like the rest of our bodies, using O2 supplied by our lungs and
blood. However, under greater exertion when the oxygen supplied by the lungs and
blood system can’t get there fast enough to keep up with the muscles’ needs, our
muscles can switch over and do lactic acid fermentation. In the process of
lactic acid fermentation, the 3-carbon pyruvic acid molecules are turned intolactic acid. It is the
presence of lactic acid in yogurt that gives it its sour taste, and it is the
presence of lactic acid in our muscles “the morning after” that makes them so
sore. Once our muscles form lactic acid, they can’t do anything else with it, so
until it is gradually washed away by the blood stream and carried to the liver
(which is able to get rid of it), our over-exerted muscles feel stiff and sore
even if they haven’t been physically injured.
Alcohol fermentation is done by yeast and some kinds of bacteria. The “waste”
products of this process are ethanol and carbon dioxide
(CO2). Humans have long taken advantage of
this process in making bread, beer, and wine. In bread making, it is the CO2 which forms and is trapped between the gluten (a
long protein in wheat) molecules that causes the bread to rise, and the ethanol
(often abbreviated as EtOH – do you remember how to draw it?) evaporating that
gives it its wonderful smell while baking. The effects of the ethanol in beer
and wine are something with which many college students are familiar (sometimes
too familiar?), and it is the CO2 produced by
the process of fermentation that makes these beverages effervescent.
Cellular Respiration:
 |
|
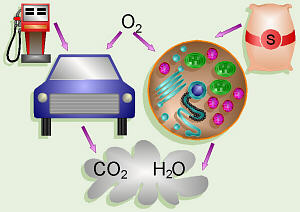
An analogy can be drawn between the process of cellular respiration in
our cells and a car. The mitochondria are the engines of our cells where
sugar is burned for fuel and the exhaust is CO2 and H2O. Note that in a car that burned fuel
perfectly, the only exhaust should theoretically be CO2 and H2O
also.
There are three steps in the process of cellular respiration:
glycolysis, theKrebs cycle, and theelectron
transport chain. |
|
In contrast to fermentation, in the process of cellular
respiration, the pyruvic acid molecules are broken down completely to
CO2 and more energy released. Note that
three molecules of O2 must react with
each molecule of pyruvic acid to form the three carbon dioxide molecules,
and three molecules of water are also formed to “use up” the hydrogens. As
mentioned above, in glycolysis, a total of four molecules of ATP are
produced, but two are used up in other steps in the process. Additional
ATP is produced during the Krebs Cycle and the Electron Transport Chain,
resulting in a grand total of 40 ATP molecules produced from the breakdown
of one molecule of glucose via cellular respiration. Since two of those
are used up during glycolysis, in prokaryotes a net total of 38
molecules of ATP are produced by cellular respiration. Most prokaryotes
have very simple cells which lack several types of organelles present in
eukaryotes, and therefore the Krebs Cycle and the Electron Transport Chain
occur in the cytoplasm and/or using chemicals embedded in the cell
membrane. In contrast, eukaryotes have more complex cells with more
specialized organelles to perform given functions. In eukaryotes, the
Krebs Cycle and Electron Transport Chain occur within the mitochondria,
and thus the pyruvic acid resulting from glycolysis must be sent into the
mitochondria for these reactions to occur. However, to move one molecule
of pyruvic acid (remember each molecule of glucose turns into two pyruvic
acid molecules) from the cytoplasm into a mitochondrion “costs” the cell
one molecule of ATP (therefore two ATPs for a whole glucose), thus a
net total of 36 ATP molecules per molecule of glucose is produced
in eukaryotes as compared to only two in fermentation. The overall
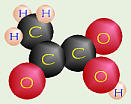
reaction for cellular respiration is C6H12O6 + 6O2  6CO2 + 6H2O (+ energy for the cell to use for
other things). 6CO2 + 6H2O (+ energy for the cell to use for
other things). |
|
 |
| Pyruvic Acid + 2 H+ |
| + 3 O2 |
 |
 |
 |
 |
| 3 Carbon Dioxide |
+ 3 H2O
+ 34
ATP |
In glycolysis and the Krebs cycle, there are also a
number of electrons released as the glucose molecule is broken down. The cell
must deal with these electrons in some way, so they are stored by the cell by
forming a compound calledNADH
by the chemical reaction, NAD+ + H+ + 2e–
 NADH. This
NADH is used to carry the electrons to the electron transport chain,
where more energy is harvested from them.
NADH. This
NADH is used to carry the electrons to the electron transport chain,
where more energy is harvested from them.
In eukaryotes, the pyruvic acid from glycolysis must be transferred into the
mitochondria to be sent through the Krebs cycle, also known as thecitric acid
cycle, at a “cost” of one ATP per molecule of pyruvic acid. In this
cycle, discovered by Hans Krebs, the pyruvic acid molecules are converted to
CO2, and two more ATP molecules are produced
per molecule of glucose. First, each 3-carbon pyruvic acid molecule has a CO2 broken off and the other
two carbons are transferred to a molecule calledacetyl coenzyme
A, while a molecule of NADH is formed from NAD+ for each pyruvic acid (= 2 for the whole
glucose). These acetyl CoA molecules are put into the actual cycle, and after
the coenzyme A part is released, eventually each 2-carbon piece is broken apart
into two molecules of CO2. In the process,
for each acetyl CoA that goes into the cycle, three molecules of NADH, one
molecule of FADH2, and one molecule of ATP
are formed (= 6 NADH, 2 FADH2, and 2 ATP per
whole glucose).
 |
|
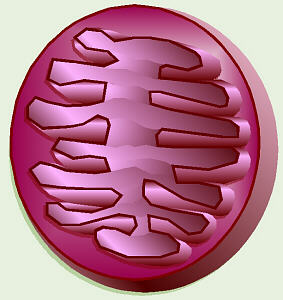
The electron transport chain is a system of
electron carriers embedded into the inner membrane of amitochondrion. As
electrons are passed from one compound to the next in the chain, their
energy is harvested and stored by forming ATP. For each molecule of NADH
which puts its two electrons in, approximately three molecules of ATP are
formed, and for each molecule of FADH2,
about two molecules of ATP are formed. |
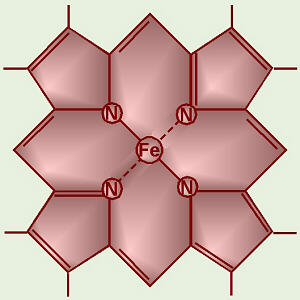
Many of the compounds that make up the electron
transport chain belong to a special group of chemicals calledcytochromes. The central structure of a
cytochrome is aporphyrin ring like
chlorophyll but with iron in the center (chlorophyll has magnesium). A
porphyrin with iron in the center is called aheme group, and
these are also found in hemoglobin in our blood.
At the last step in the electron transport chain, the “used up”
electrons, along with some “spare” hydrogen ions are combined with O2 (we finally got around to the O2) to form water as a waste product: 4e- + 4H+ + O2  2H2O. 2H2O. |
|
Click on the heme group
to see how to draw one. |
 |
|
| |
Many of the enzymes in the cells of organisms need other
helpers to function. These non-protein enzyme helpers are calledcofactors and can include substances like iron, zinc,
or copper. If a cofactor is an organic molecule, it then is called acoenzyme. Many of the vitamins needed
by our bodies are used as coenzymes to help our enzymes to do their jobs.
Vitamin B1(thiamine) is a coenzyme used in
removing CO2 from various organic compounds.
B2(riboflavin) is a component ofFAD (or
FADH2), one of the chemicals used to
transport electrons from the Krebs cycle to the electron transport chain.
Vitamin B3(niacin) is a component of
NAD+ (or NADH) which is the major
transporter of electrons from glycolysis and the Krebs cycle to the electron
transport chain. Without enough of these B vitamins, our ability to get the
energy out of our food would come to a grinding halt! B6(pyridoxine), B12(cobalamin),pantothenic acid,folic acid, andbiotin are all other B vitamins
which serve as coenzymes at various points in metabolizing our food.
Interestingly, B12 has cobalt in it, a
mineral which we need in only very minute quantities, but whose absence can
cause symptoms of deficiency.
My mother once had a friend who hadporphyria, a dominant genetic
disorder in which the person’s body cannot properly make porphyrin rings. This
would, thus, affect the person’s ability to make both hemoglobin to carry oxygen
in the blood and cytochromes for the electron transport chain. This woman’s
symptoms were quite variable. At times, she would appear nearly normal while on
other occasions she would have to be hospitalized for temporary paralysis of
part of her body or other symptoms. There were a number of foods and drugs she
had to avoid because they would trigger or worsen her symptoms. She frequently
was in a lot of pain. Because porphyria is a dominant genetic disorder, there
was a 50% chance this woman’s daughter would also have porphyria. Thus after the
woman was diagnosed with porphyria, a number of tests were also run on the girl,
and she was more carefully monitored as she grew up. My mother eventually lost
contact with them, so I never heard the end of the story.
Because there are a number of enzymes and steps involved in forming porphyrin
rings, there are a number of possible points in the process where genetic
defects could occur. The Merck Manual says there are eight steps in the process
of making porphyrin rings, with genetic abnormalities possible in seven of the
eight enzymes.
Several years ago, Dr. Fankhauser mentioned to me that he heard somewhere
that an “average” 70 kg (= 154 lb) person makes about 40 kg (= 88 lb) of
ATP/day, which would be 57% of that person’s body weight. As we discussed that,
the question arose, “What would be the maximum amount of ATP that a person could
possibly make?” To try to come up with an answer to that question, I did the
following calculations.
- First, let’s assume that person eats an “average” dietary intake of 2500
KCal of food energy (a number listed on the side of many food packages and a
reasonable amount that such a person might consume).
- However, just out of curiosity, let’s assume that all (100%) of that is
glucose (In real life, that would be a terrible idea! We need all the other
nutrients that we get from eating a variety of foods.). Since carbohydrates
store about 4 KCal of energy per gram, that would mean that 2500 KCal of
glucose would be equivalent to 625 g (= 1.4 lb) of glucose. Since the
molecular weight of glucose is 180 g/m, this would be equivalent to 3.47 moles
of glucose.
- Also, just for the sake of argument, let’s assume that 100% of the
ingested glucose is burned for fuel, and that the process is 100% efficient so
there is no waste (in real life, our bodies would never use all 100% for fuel
– some gets used to build other chemicals, and just like the fuel efficiency
in our automobiles, the process is never 100% efficient.). Since, as was
mentioned above, eukaryotes make about 36 moles of ATP from every mole of
glucose, then those 3.74 moles of glucose would be equivalent to 125 moles of
ATP.
- The molecular weight of ATP is 507 g/m, so that would
be 63375 g or 63.375 kg of ATP.
- Thus, if it was really possible to meet all of those background
assumptions and a 70 kg person really could make 63 kg of ATP, that would be
90% of that person’s body weight! However, to think that we make even 57% –
about half – of our body weight each day in ATP is pretty amazing.
As another example:
- suppose a person would consume one 12-oz. can of soft drink,
- most types of soft drink contain about 41 to 49 g of sugar, so let’s say
this soft drink contains 45 g,
- suppose all of that sugar would be glucose,
- suppose the person’s body burns all of that sugar for fuel and does not
store any of it as fat or use any of it in other ways, and
- suppose the process of cellular respiration is 100% efficient and the
sugar is completely oxidized to CO2 and H2O.
Then:
- since the molecular weight of glucose is 180g/m, the 45 g of glucose
would be 0.25 m,
- since cellular respiration produces 36 m ATP for each 1 m of glucose,
that would make 9 m of ATP, and
- since the MW of ATP is 507 g/m, that would be equivalent to
4563 g (about 10 lb) of ATP.
Recently I received an e-mail message from a student who asked how long the
whole process takes. While I have never seen any information on that in print, a
rough approximation can also be calculated from the above statistic:
- If, as mentioned above, an “average” 70 kg person makes about 40 kg of
ATP/day, then
40 kg/24 hr × 1 hr/60 min × 1000 g/kg = about 27.8 g/min.
- Since the molecular weight of ATP is 507 g/m, then
that
27.8 g/min × 1 m/507 g = 0.0548 m/min.
- Avagadro’s number says that there are always 6.02 x 1023 molecules/mole,
so 0.0548 m/min × 6.02 x
1023 molecules/mole = 3.30 x 1022 molecules/min.
- or, since there are 60 sec/min, then that’s
3.30 x 1022 molecules/min × 1min/60 sec = 5.50 x 1020 molecules/sec made by a 70 kg body.
- so that would be equivalent to
5.50 x 1020 molecules/sec ÷ 70 kg = 7.85 × 1018 molecules/sec/kg of body
- or × 1kg/1000 g = 7.85 × 1015
molecules/sec/g of body
- or × 1g/1000 mg = 7.85 × 1012
molecules/sec/mg of body
- or × 1mg/1000 µg = 7.85 × 109
molecules/sec/µg of body.
References:
- Berkow, Robert, ed. 1999. The Merck Manual. 17th ed. Merck, Sharp &
Dohme, Rahway, NJ.
- Borror, Donald J. 1960. Dictionary of Root Words and Combining Forms.
Mayfield Publ. Co.
- Campbell, Neil A., Lawrence G. Mitchell, Jane B. Reece. 1999. Biology,
5th Ed. Benjamin/Cummings Publ. Co.,
Inc. Menlo Park, CA. (plus earlier editions)
- Campbell, Neil A., Lawrence G. Mitchell, Jane B. Reece. 1999. Biology:
Concepts and Connections, 3rd Ed.
Benjamin/Cummings Publ. Co., Inc. Menlo Park, CA. (plus earlier editions)
- Marchuk, William N. 1992. A Life Science Lexicon. Wm. C. Brown Publishers,
Dubuque, IA.